CasNo: 811-97-2
Molecular Formula: C2H2F4
Appearance: colourless gas or cryogenic liquid
|
Physical and Chemical Properties |
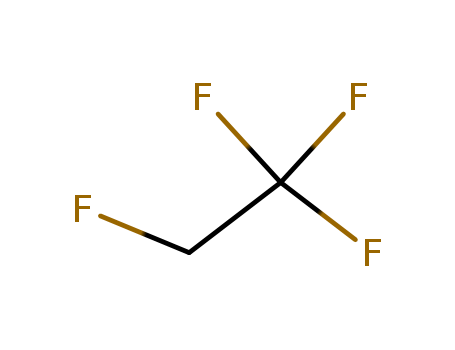
1, 1, 1, 2-Tetrafluoroethane is commonly known as R134a, HFC134a and HFC-134a. It is a kind of colorless, non-toxic and non-burning chemical. It is insoluble in water (67mg/L, 25 ℃ ) but soluble in ether with its potential value of ozone depletion being 0. Its thermodynamic property is very similar as CFC-12 while having its security being comparable to CFC-12, and thus has been recognized as the best substitute of CFC-12. Although there are some greenhouse effect for HFC-134a (HGWP = 0.28), this doesn’t affect it to become the primary-choice ODS (Ozone Depleting Substances) substitute. 1, 1, 1, 2-tetrafluoroethane (HFC-134a or HFA-134a) is a new generation of non-chlorofluorocarbon compounds as pharmaceutical excipients. It is mainly used as the propellant agent contained in the mist agent during the treatment of asthma and chronic respiratory disorders disease. Compared with the traditional CFC class pharmaceutical propellant, the advantage of HFC-134a is free of chlorine atom and thus having zero ODP (ozone depletion potential) value and GWP value (global warming potential) without depleting ozone and generating photochemical smog and is chemically inert and toxicologically safe. It is also a kind of environmental friendly pharmaceutical excipients and is also currently used as the major substitute of CFC contained in aerosol that is ozone-depleting. Figure 1 is the chemical structure formula of tetra-fluoroethane. |
|
Environmental friendly refrigerants |
Tetrafluoroethane (R-134a) is the most widely used low or moderate-temperature refrigerant. Owing to the excellent overall performance of the tetrafluoroethane (HFC-134a), it has become a very effective and safe substitute for CFC-12 products. It is mainly applied to various areas taking advantage of R-12 (R12, Freon 12, F-12, CFC-12, Freon 12, dichlorodifluoromethane) refrigerant including: refrigerators, freezers, water dispensers, auto air conditioning, central air conditioning, dehumidifiers, cold storage, commercial refrigeration, ice machines, ice cream machine, refrigeration condensing units and other refrigeration equipment. It can also be applied to fields of aerosol propellants, medical aerosols, pesticides propellant, polymer (plastic) physical foaming agent, and protection gas of magnesium alloy. While tetrafluoroethane refrigerant (R-134a) is the most popular choice as alternative of the feron R12 for being applied to the newly installed refrigeration equipment, owing to that R134a is different from R12 in physical and chemical properties, theoretical cycle performance as well as the applied compressor oil, for the after-sales repair of the refrigerated equipment with initial installation of R12 refrigerant refrigeration equipment repairs, if you need to add or replace the refrigerant, you have no choice but still add R12. Usually people can‘t directly apply tetrafluoroethane refrigerant (R-134a) to replace R12 (That is usually called “no cataclysmic replacement”). |
|
Synthetic route |
Synthetic route of the raw material of 1, 1, 1, 2-tetfluoroethane: it has been reported of as much as several dozens of major synthetic routes. Among them, the major synthetic route is shown as the figure. For various synthetic routes, considering comprehensively of the sources of raw materials, production processes and waste treatment and other factors, only two routes of raw materials, trichlorethylene and tetrachlorethylene have practical value of industrial production. In the actual industrial production, trichlorethylene raw material routes, due to its simple reaction step and small amounts of by-products, is preferentially recommended. Trichlorethylene route, the main production process are liquid, gas and gas-liquid method. Take trichlorethylene (TCE) and hydrogen fluoride (HF) as raw materials, upon the action of catalyst, perform addition and substitution reaction in the first step to generate 1,1,1-trifluoro-2-chloroethane (HCFC-133a ); then, at higher temperatures, perform the second step to generate 1,1,1,2-tetrafluoroethane (HFC-134a). The reaction equation is as follows: The advantage of liquid Freon is following the traditional production methods with simple production process and relatively mature technology. In 1982, DuPont ha applied liquid fluorination for the manufacturing of HFC-134a. However, at high temperature, due to the emergent corrosion of the equipment and the difficulty in conducting continuous production, this method is still in the stage of small-range laboratory test. Shanghai Institute of Organic Chemistry Research has applied Cl (CF2CF2) 4OCF2SO2F (perfluoroalkoxy sulfonyl fluoride) as a catalyst and have reaction in the KF solution at the pressure of 230e and 12.5MPa for 2h to give HC-134a with the yield being 88%. The reaction equation is: CF3CH2Cl + KF---CF3CH2F + KCl, compared with the DuPont method, the CAS Shanghai Institute of Organic Chemistry had achieved lower reaction temperature so that corrosion and byproducts have been effectively controlled, making it possible to conduct the continuous production. However, it is still difficult to achieve industrial production using this method in short term. The above information is edited by the lookchem of Dai Xiongfeng. |
|
Gas - liquid phase and gas-phase synthesis of tetra-fluoroethane |
The advantage of gas-Liquid method is that at the first-step reaction, it can almost take advantage of all the equipment and technology, liquid-phase washing, alkaline washing and drying processes for the original production of Freon products. This can effectively reduce the energy consumption. This process, for the old plant of the original production of CFCs, it is a doable route. However, the second step is equilibrium reaction with low gas one-way conversion rate and short duration life of the catalyst and other shortcomings. Therefore, this step restricts the vapor-liquid phase process for being applied to process route for large-scale production. Gas phase method applies trichlorethylene (TCE) and anhydrous hydrogen fluoride (HF) for reaction in the action of a chromium-containing catalyst. The first step of addition and substitution reactions generates HCFC-133a, and then it is further reacted with HF in the presence of chromium-based catalyst to generate the finished product, tetrafluoroethane (HFC-134a) at a temperature of 350~380 ℃. The second-stage reaction of gas-phase method is relative difficult with the conversion rate being generally only about 20%. Therefore, in the industrial production, people mostly adopts continuous cycle method to have the large amount of raw materials be recycled to reduce the toxic and hazardous intermediate products as well as improve the overall yield. Gas-phase method has a lot of advantages including easily controllable reaction process, small amount of waste pollution and easily being applied for large-scale continuous production. Currently gas-phase method has gradually replaced liquid-phase method and gas phase-liquid phase method to become the mainstream of the world's production of tetrafluoroethane (HFC-134a). |
|
Precautions for manipulation |
Technical measures: it should be manipulated in a well-ventilated place. Upon high pressure condition, make sure that the internal pressure of the reaction apparatus does not exceed the cylinder pressure. For safety purpose, the gas flow path should be installed with a check valve. Do not remove the check valve before running out of the content. Wear protective equipment when handling. Wash hands and face thoroughly after handling. Handling Precautions: Avoid contact with skin, eyes and clothing. Storage conditions: Avoid the sunshine. Store it in a well-ventilated place. Do not expose it to environment above 40 ℃. Locked up the place where it is stored. Store it away from incompatible materials such as oxidants. |
|
Production Methods |
Tetrafluoroethane can be prepared by several different routes; however, the following routes of preparation illustrate the methods used: Isomerization/hydrofluorination of 1,1,2-trichloro-1,2,2-trifluoroethane (CFC-113) to 1,1-dichloro-1,2,2,2-tetrafluoroethane (CFC-114a), followed by hydrodechlorination of the latter. Hydrofluorination of trichloroethylene, via 1-chloro-1,1,1- trifluoroethane (HCFC-133a). |
|
Air & Water Reactions |
Insoluble in water. |
|
Reactivity Profile |
1,1,1,2-Tetrafluoroethane is chemically inert in many situations, but can react violently with strong reducing agents such as the very active metals and the active metals. Can react with strong oxidizing agents or weaker oxidizing agents under extremes of temperature. |
|
Flammability and Explosibility |
Nonflammable |
|
Pharmaceutical Applications |
Tetrafluoroethane is a hydrofluorocarbon (HFC) or hydrofluoroalkane (HFA) aerosol propellant (contains hydrogen, fluorine, and carbon) as contrasted to a CFC (chlorine, fluorine, and carbon). The lack of chlorine in the molecule and the presence of hydrogen reduce the ozone depletion activity to practically zero. Hence tetrafluoroethane is an alternative to CFCs in the formulation of metereddose inhalers (MDIs). It has replaced CFC-12 as a refrigerant and propellant since it has essentially the same vapor pressure. Its very low Kauri-butanol value and solubility parameter indicate that it is not a good solvent for the commonly used surfactants for MDIs. Sorbitan trioleate, sorbitan sesquioleate, oleic acid, and soya lecithin show limited solubility in tetrafluoroethane and the amount of surfactant that actually dissolves may not be sufficient to keep a drug readily dispersed. Up to 10% ethanol may be used to increase its solubility. When tetrafluoroethane (P-134a) is used for pharmaceutical aerosols and MDIs, the pharmaceutical grade must be specified. Industrial grades may not be satisfactory due to their impurity profiles. |
|
Safety |
Tetrafluoroethane is used as a refrigerant and as a non-CFC propellant in various aerosols including topical pharmaceuticals and MDIs. Tetrafluoroethane is regarded as nontoxic and nonirritating when used as directed. No acute or chronic hazard is present when exposures to the vapor are below the acceptable exposure limit (AEL) of 1000 ppm, 8-hour and 12-hour time weighed average (TWA). In this regard it has the same value as the threshold limit value (TLV) for CFC-12. Inhaling a high concentration of tetrafluoroethane vapors can be harmful and is similar to inhaling vapors of CFC-12. Intentional inhalation of vapors of tetrafluoroethane can be dangerous and may cause death. The same labeling required on CFC aerosols would be required for those containing tetrafluoroethane as a propellant (except for the EPA requirement). |
|
Carcinogenicity |
The results from three lifetime inhalation carcinogenesis studies with HFC 134a have been published. The first one involved exposure of groups of 80 male and 80 female rats to levels of ≤50,000 ppm 6 h/ day, 5 days/week for 2 years.An increase inLeydig cell tumors was seen in themale rats at 50,000 ppm(30%) compared to the air-exposed controls (12%). Likewise, therewas an increase in Leydig cell hyperplasia. No effects were seen at 10,000 ppm (370). The second study with rats involved snout-only inhalation exposures to levels of ≤50,000 ppm 1 h/day, 7 days/week for 108 weeks. The same investigators conducted a lifetime study withmice. In this study, groups of mice were exposed to snout-only levels of ≤75,000 ppm 1 h/day, 7 days/week for 104 weeks. No adverse effects were seen in either rats or mice. Since the total dose received by the rats in the high exposure level of this study was lower than in the Collins’ study, this report supports the observation that 10,000 ppm, 6 h/day, 5 days/week for 2 years was a NOEL. Rats were given 300 mg of HFC 134a in corn oil 5 days/ week for 52 weeks and held for a total of 125 weeks. There was no evidence for carcinogenicity. |
|
storage |
Tetrafluoroethane is a nonreactive and stable material. The liquified gas is stable when used as a propellant and should be stored in a metal cylinder in a cool dry place. |
|
Incompatibilities |
The major incompatibility of tetrafluoroethane is its lack of miscibility with water. Since it has a very low Kauri-butanol value, tetrafluoroethane is considered to be a very poor solvent for most drugs used in MDI formulations. It also shows a low solubility for some of the commonly used MDI surfactants. |
|
Regulatory Status |
Included in the FDA Inactive Ingredients Database (aerosol formulations for inhalation and nasal applications). Included in nonparenteral medicines licensed in the UK. |
|
General Description |
A colorless gas with a slight ethereal odor. Vapors are heavier than air. Shipped liquefied under own vapor pressure. Flash point 351°F. Inhalation at high concentrations is harmful and may cause heart irregularities, unconsciousness or death without warning. Liquid contact may cause frostbite. Vapors can replace the available oxygen. |
InChI:InChI=1/C2H2F4/c3-1-2(4,5)6/h1H2
-
The microwave spectrum of CF3CH2F has be...
The dechlorination of 2-chloro-1,1,1,2-t...
Different fluorinated catalysts based on...
A series of CrOx-Y2O3 catalysts were pre...
Efficient chemistry is described for the...
PROBLEM TO BE SOLVED: To provide a novel...
A catalyst comprising one or more metal ...
A catalyst comprising chromia and at lea...
The invention discloses a device for pre...
1,1,1-trifluoro-2-chloroethane

2,2,2-trifluoroethanol

1,1,1,2-tetrafluoroethane

1-Chloro-2,2-difluoroethene

1,1,1-trifluoro-2-(2,2,2-trifluoroethoxy)ethane
| Conditions | Yield |
|---|---|
|
With potassium hydroxide; at 250 ℃; for 13h; under 77520 Torr;
|
70% |
chloroform

pentaerythrityl tetrachloride

1,1,1,2-tetrafluoroethane

1,1,1-trifluoro-2-chloroethane

1,2-dichloro-1,1-difluoroethane

1,2,3-trichloro-2-chloromethyl-propane

1,1,2,2-tetrachloroethane
| Conditions | Yield |
|---|---|
|
With fluorinated gallium(III) oxide; Product distribution; Ambient temperature; further reagent: fluorinated indium(III) oxide;
|
2,2,2-Trifluoroethyl p-toluenesulfonate
glycolic Acid
Vinylidene fluoride
Fluoroacetic acid
Carbonyl fluoride
formyl fluoride
trifluoroacetyl fluoride
trifluoromethanol