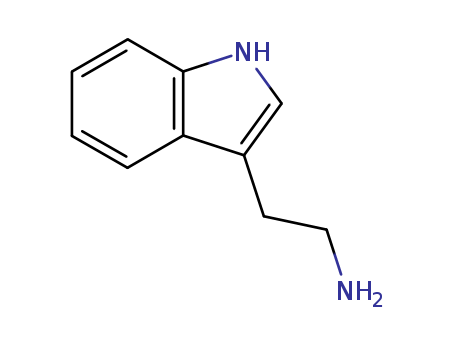
CasNo: 61-54-1
Molecular Formula: C10H12N2
Appearance: White to orange crystalline powder
|
Plants Containing Tryptamine |
In plants, tryptamine in small amounts acts as a promising phase to the plant hormone indole-3-acetic acid in one biosynthetic pathway. N, N-dimethyltryptamine (DMT) is a tryptamine derivative that is an active constituent for the hallucinogenic effect of brew known as the “vine of the souls.” Indigenous Amazonian tribes have traditionally used the drink for therapeutic purposes for effective treatment of some physical maladies and abuse disorders. Magic mushrooms are the most common fungi that contain tryptamine derivatives. |
|
Pharmacology |
Serotonin (5-hydroxytryptamine, 5-HT), which is a natural derivative of tryptamine is a significant signaling hormone that helps in the modulation and regulation of numerous processes within the central nervous system, for instance, cognition, sleep, temperature regulation, memory, and behavior. The mammalian brain contains small traces of tryptamine, which generally act as a modulator or neurotransmitter by releasing serotonin agents. It is also an enhancer of serotonergic activity. Only one minute is required for tryptamine to produce psychotropic phenomena when used recreationally. It has been associated with fatalities and intoxications for multiple reasons including low toxic concentrations. |
|
Preparation |
Tryptamine, a monoamine alkaloid containing an indole ring structure is derived by the decarboxylation of amino acid tryptophan.The synthesis of tryptamines is typically conducted following a classic route starting with a Mannich reaction of an indole heterocycle, followed by quaternization of the amine, nucleophilic substitution with highly toxic cyanide and final reduction. |
|
Biochem/physiol Actions |
Vasoactive; may have a neuromodulator function; biogenic amine formed from the decarboxylation of tryptophan by L-aromatic amino acid decarboxylase. |
|
Purification Methods |
Crystallise tryptamine from *benzene, Et2O (m 114o) or pet ether (m 118o). It has UV: 222n 276, 282 and 291nm (EtOH) and max 226, 275, 281 and 290nm (HCl). [Beilstein 22 II 346, 22 III/IV 4319, 22/10 V 45.] |
|
|
|
|
General Description |
Tryptamine is a monoamine alkaloid derived from the amino acid tryptophan, serving as a fundamental structural motif in various biologically active compounds, including neurotransmitters and psychedelic substances. It plays a role in the synthesis of inhibitors targeting cancer resistance proteins, as well as in the development of dopamine receptor ligands and alkaloid precursors. Its versatility in chemical modifications allows it to contribute to diverse pharmacological applications, such as modulating receptor affinities or serving as a scaffold for complex natural product synthesis. |
|
Applications |
Analogs of tryptamine that are typically produced by its synthetic modification play a significant role in individuals due to the introduction of functionalities that are biologically active in its nucleus that may cause changes in the mental and physical status of the human brain. Substitutions on the indole ring at nitrogen and C-2 of its side chain produce numerous neuroactive compounds ranging from anti-migraine drugs to toxic substances, such as rizatriptan, sumatriptan, and zolmitriptan. A small amount of tryptamine is required due to its fatalities and intoxication for several reasons. |
|
Definition |
ChEBI: Tryptamine is an aminoalkylindole consisting of indole having a 2-aminoethyl group at the 3-position. It has a role as a human metabolite, a plant metabolite and a mouse metabolite. It is an aminoalkylindole, an indole alkaloid, an aralkylamino compound and a member of tryptamines. It is a conjugate base of a tryptaminium. |
InChI:InChI=1/C10H12N2/c11-6-5-8-7-12-10-4-2-1-3-9(8)10/h1-4,7,12H,5-6,11H2
-
A series of N-skatyltryptamines was synt...
Two series of naphthoquinone and anthraq...
Most of the previous syntheses of the ma...
We previously identified a spiro [piperi...
naproxen

Trp

tryptamine

quinolin-4-ol
| Conditions | Yield |
|---|---|
|
With Saccharomyces cerevisiae wild type strain V328; for 0.333333h; UV-irradiation;
|
methylene blue

Trp

tryptamine

quinolin-4-ol

2-amino-4-(2-formamidophenyl)-4-oxo-butanoic acid

Kynurenine
| Conditions | Yield |
|---|---|
|
With Saccharomyces cerevisiae wild type strain V328; for 0.333333h; UV-irradiation;
|
indole-3-acetonitrile
3-(2-bromoethyl)-1H-indole
3-indolylacetamide
3-[(E)-2-nitroeth-1-enyl]-1H-indole
N-[2-(3-indolyl)ethyl]nicotinamide
15,16,17,18,19,20-hexahydroyohimban-21-one
9H-benzo[c]indolo[3,2,1-ij][1,5]naphthyridin-9-one
N-phthalimidotryptamine