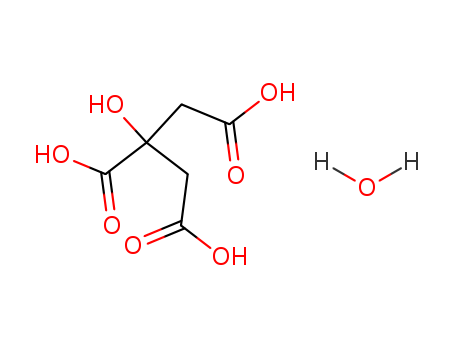
CasNo: 5949-29-1
Molecular Formula: C6H10O8
Appearance: white crystals or powder
| Description | Citric acid monohydrate is a multifunctional compound widely utilized in the food industry for its preservative and acidity-regulating properties. Its applications extend to pharmaceuticals and dietary supplements, where it serves as an excipient with antioxidant properties, contributing to stability and safety. The versatility of citric acid monohydrate makes it a valuable component in various industries. |
| Uses | Used as an excipient in pharmaceutical preparations. Acts as an antioxidant, maintaining stability of active ingredients. Functions as a preservative. |
| Applications in Food Industry | Used as a preservative in the food industry. Regulates acidity and pH levels in food products. Used in the preparation of lemon pepper seasoning. Ice cream manufacturers use it as an emulsifying agent to prevent fat separation and sucrose crystallization. Can be used in recipes as a substitute for fresh lemon juice. |
| Form | Non-toxic, with low reactivity. Very soluble in water. Occurs as colorless crystals or as a white, crystalline powder with a strongly acidic taste. |
| Odor and Taste | Odorless with a strong acidic taste. |
| Crystal Structure | Orthorhombic |
| Solubility | Highly soluble in water. |
| Acidulant | Used as an acidulant in various food products to control pH. |
| Toxicity and Reactivity | Non-toxic. Has low reactivity. |
| Flavoring Agent | Adds a sour taste to candies, cookies, biscuits, canned fruits, jams, and jellies. |
| Antistaling Agent | Used as an antistaling agent in bakery products. |
| Excipient in Pharmaceuticals | Used in pharmaceutical preparations as an excipient due to its antioxidant properties, helping maintain the stability of active ingredients. |
InChI:InChI=1/C6H8O7.H2O/c7-3(8)1-6(13,5(11)12)2-4(9)10;/h13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);1H2
An experimental study concerned with the growth kinetics of citric acid monohydrate crystals is presented. Due to the properties of the system, which is very soluble, with low solid-liquid …
The knowledge of the thermodynamic parameters related to the hydration of organic solid phases is paramount to exert a very good control over this transition during industrial production or storage.
The objective of the study was to synthesize deep eutectic solvents (DES) using choline chloride and citric acid monohydrate at different molar ratios (i.e. DES 3:1, 2:1, 1:1, 1:2 and 1:3) followed by physicochemical characterization.
sodium chlorite

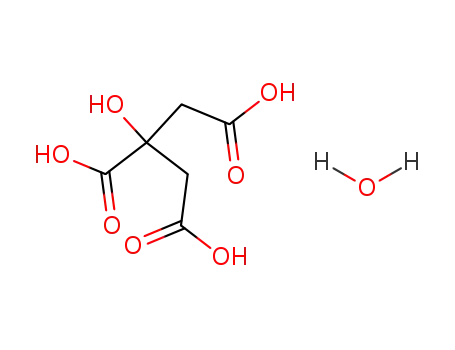
citric acid monohydrate
| Conditions | Yield |
|---|---|
|
With sodium hydroxide;
|


citric acid monohydrate

sodium chlorite
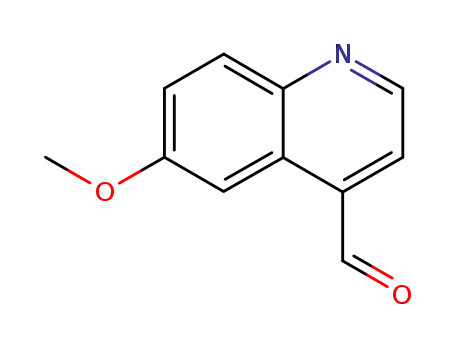
6-methoxyquinoline-4-carbaldehyde
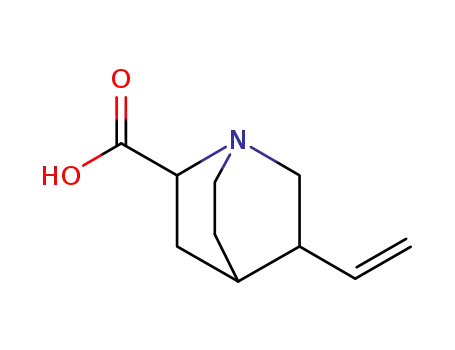
5-vinyl-1-aza-bicyclo[2.2.2]octane-2-carboxylic acid
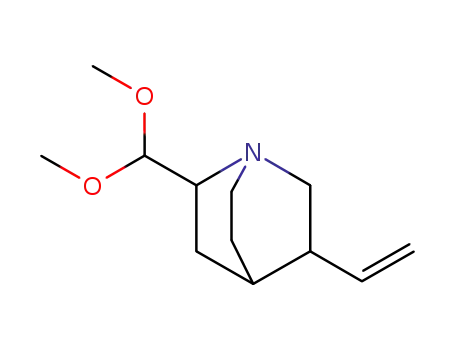
2-Dimethoxymethyl-5-vinyl-1-aza-bicyclo[2.2.2]octane
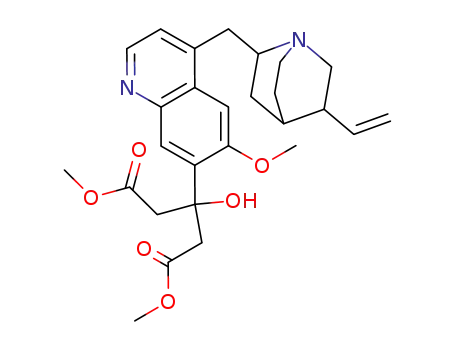
7'-(1,3-dimethoxycarbonyl-2-hydroxyprop-2-yl)-deoxyquinine